 
NO molecule: Although NO is a diatomic molecule, it is possible to draw the Lewis structure by following the procedure in section 1.2.2. Examples could include NO (nitrogen monoxide or nitric oxide), NO2 (nitrogen dioxide) and alkyl radicals. If the total number of valence electrons is an odd number, the octet rule can not be applied to all atoms in the species. Here, we will see some other cases in which the octet rule is compromised. So far, we have always been applying the octet rule in Lewis structures however, there are some cases in which this rule does not apply. All of the atoms (except H) should have 8 electrons around it, therefore, N usually has 1 lone pair, O has 2 lone pairs and halogens have 3 lone pairs.ġ.2.5 Exceptions to the Octet Rule in Lewis Structure To count how many lone pairs should be involved on a certain atom, apply the octet rule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
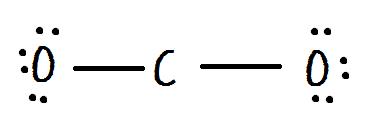
 RSS Feed
RSS Feed
